- History
- Institutions

A public listed company ,
Jiangsu Nanno antibody R&D center created by joint collaboration with South-East University, awarded as the Nantong branch of national level post-doctoral R&D station
Awarded as the High-tech enterprise in China
Acquired ISO 9001(TUV) ISO 14001,ISO9001, ISO13458
HBV-5 Rapid Tests certified by sFDA
EGENS invested in KEWEI Diagnostics
HBV-5 Rapid Tests developed with 5 IPRs
Nantong Egens Biotechnology Co.,Ltd established, the first IVD manufacture in Jiangsu Province,P.R.China
1000 sqm2 GMP production facility established
The first manufacture with HEV antibody Elisa Test kits, papers published in Chinese Journal of Laboratory Medicine & Chinese Journal of Experimental and Clinical Virology
The first manufacture certified for HCV antibody Elisa Test Kits production by sFDA.
HAV IgM antibody Elisa Test Kits developed and implemented, awarded th 2nd prize of the scientific progress by the P.L.A.
Certified as the Quality Control Centre of Infectious disease Reagent by the Ministry of Health of Medical Department of Ceneral Logistics Department.
Established “Beijing Kewei Clinical Diagnostic Reagent, Inc.” Implemented project of developing Elisaimmunoassay (co-projector: the military hospital 302), the first one with HBV series of Elisa Test kits.
-
2016
01 -
2014
12 -
2013
08 -
2010
10 -
2007
12 -
2006
08 -
2003
12 -
2002
03 -
1999
12 -
1998
08 -
1997
06 -
1995
03 -
1992
07 -
1992
05 -
1990
03

2014


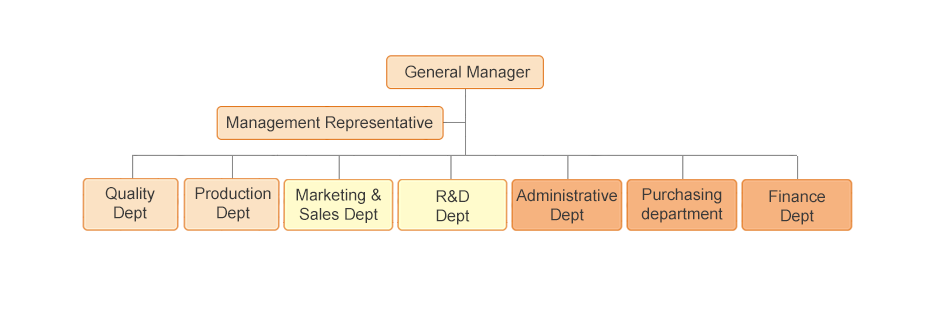
EGENS Organization Structure